$ 0.000 6.69%
Photon (PHO) Rank 3909
Photon is a Cryptocurrency using the Blake 256 Algorithm. It is a new project designed to function well now and with future technology in mind. The Blake-256 algorithm hash rate is just under 3x faster on the GPU and just over 2x on the FPGA compared with Bitcoin The reward for mining Photon does NOT decrease over time, instead it increases with block height and difficulty There are no restrictions on any platform as Photon does NOT include artificial *Security* or *Resistance* that reduce mining hash rate and power efficiency
| Mkt.Cap | $ 0.00000000 | Volume 24H | 0.00000000PHO |
| Market share | 0% | Total Supply | 90 BPHO |
| Proof type | PoW | Open | $ 0.0003 |
| Low | $ 0.0003 | High | $ 0.0003 |
How much does light weigh?
photon - Computer Definition

The range of photon energies for visible light from red to violet is 1.63 to 3.26 eV, respectively (left for this chapter’s Problems and Exercises to verify). These energies are on the order of those between outer electron shells in atoms and molecules.

Do photons have energy?
If by "matter" you mean massive (in the sense of having nonzero rest mass), then photons are not matter. Photon is a fundamental particle, a force carrier for electromagnetic interaction. It has zero charge, zero rest mass, and is a spin 1 particle (a boson).

X rays are ideal for medical imaging, their most common use, and a fact that was recognized immediately upon their discovery in 1895 by the German physicist W. C. Roentgen (1845–1923). (See Figure 2.) Within one year of their discovery, x rays (for a time called Roentgen rays) were used for medical diagnostics. Roentgen received the 1901 Nobel Prize for the discovery of x rays.
Electromagnetic Field Theory. Cambridge University Press. The electron microscope directs a focused beam of electrons at a specimen. Some electrons change their properties, such as movement direction, angle, and relative phase and energy as the beam interacts with the material. However, electron microscopes are expensive instruments that are costly to maintain.
10. wave
What is the antonym for photon?
The energy of a photon depends on its wavelength: longer wavelength photons have less energy and shorter wavelength photons have more. Red photons, for example, have less energy than blue ones. More energetic wavelengths such as blue and ultraviolet caused more electrons to be ejected than red or infrared wavelengths.
The quantum energy of different photons is often used in cameras, and other machines that use visible and higher than visible radiation. This because these photons are energetic enough to ionize atoms. The theory that states that light behaves both like a wave and a particle is called the wave-particle duality theory. Photons are not thought to be made up of smaller particles.
Each of the photon's properties listed above can be exploited to carry information. For instance, human eyes, conventional cameras, and traditional space telescopes extract photon position and frequency (color) information from a group of photons in order to form images. Radio antennas vary the frequency (FM) or the photon count (AM) along the length of the radio waves that they create in order to encode information. Interferometers such as used in some space telescopes measure the phase properties of the photons in a beam to extract information about the source that created the beam.
The photon concept has led to momentous advances in experimental and theoretical physics, including lasers, Bose–Einstein condensation, quantum field theory, and the probabilistic interpretation of quantum mechanics. It has been applied to photochemistry, high-resolution microscopy, and measurements of molecular distances. Recently, photons have been studied as elements of quantum computers, and for applications in optical imaging and optical communication such as quantum cryptography.
A sodium atom has 11 electrons, and because of the way they're stacked in orbitals one of those electrons is most likely to accept and emit energy. The energy packets that this electron is most likely to emit fall right around a wavelength of 590 nanometers. This wavelength corresponds to yellow light.

Are photons smaller than atoms?
While the electron has a negative electric charge, the positron has a positive electric charge, and is produced naturally in certain types of radioactive decay. The opposite is also true: the antiparticle of the positron is the electron. Some particles, such as the photon, are their own antiparticle.
Electromagnetism is an idea that combines electricity with magnetism. One common way that we experience electromagnetism in our daily lives is light, which is caused by electromagnetism. Electromagnetism is also responsible for charge, which is the reason that you can not push your hand through a table. Since photons are the force-carrying particle of electromagnetism, they are also gauge bosons. Some matter–called dark matter–is not believed to be affected by electromagnetism.
Six of the particles in the Standard Model are quarks (shown in purple). Each of the first three columns forms a generation of matter. Despite all this complexity, there is a basic way to talk about the effective “size” of a photon, and that is through it’s wavelength.

- When visible, IR, or microwave radiation is hazardous, such as the inducement of cataracts by microwaves, the hazard is due to huge numbers of photons acting together (not to an accumulation of photons, such as sterilization by weak UV).
- The range of photon energies for visible light from red to violet is 1.63 to 3.26 eV, respectively (left for this chapter’s Problems and Exercises to verify).
- In particle physics, photons are responsible for electromagnetic force.
- Most semiconductors have a variable level of conductivity that lies between the extremes of conduction and insulation.[117] On the other hand, metals have an electronic band structure containing partially filled electronic bands.
- X rays are produced when energetic electrons strike the copper anode of this cathode ray tube (CRT).
- Violet flowers absorb red and reflect violet; this implies there is no energy step between levels in the receptor molecule equal to the violet photon’s energy, but there is an energy step for the red.
Physical properties
The Bohr model of the atom, showing states of electron with energy quantized by the number n. An electron dropping to a lower orbit emits a photon equal to the energy difference between the orbits. Lancaster, Tom; Blundell, Stephen J.; Blundell, Stephen (April 2014).
How long is a single photon?
A Photon is the Quantum of Electromagnetic Radiation while an atom is the fundamental constituent of all the Matter. Its size may be similar to the size of subatomic particles like Electron but it's smaller than an atom.
While photons don’t have a physical diameter, and can be treated as point particles, their quantum behavior gives them a probabilistic size. As a photon gets closer to another object, the chance of it interacting becomes greater. This is often represented as a cross section given in terms of area. Photon energy can be expressed using any unit of energy.
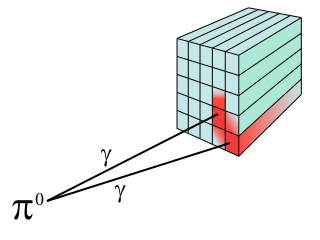
While γ rays originate in nuclear decay, x rays are produced by the process shown in Figure 3. Electrons ejected by thermal agitation from a hot filament in a vacuum tube are accelerated through a high voltage, gaining kinetic energy from the electrical potential energy. When they strike the anode, the electrons convert their kinetic energy to a variety of forms, including thermal energy. But since an accelerated charge radiates EM waves, and since the electrons act individually, photons are also produced. Some of these x-ray photons obtain the kinetic energy of the electron.

The photon is sometimes referred to as a "quantum" of electromagnetic energy. We learned on the light page that light has the unique characteristic of behaving like both a wave and a particle at the same time. We then learned some of the ways that light behaves like a wave.
Quantum Field Theory for the Gifted Amateur. Since this picture was first developed by Stueckelberg,[7] and acquired its modern form in Feynman's work,[8] it is called the Feynman–Stueckelberg interpretation of antiparticles to honor both scientists. Then the energy of the vacuum is exactly E0. Since all energies are measured relative to the vacuum, H is positive definite. Analysis of the properties of ak and bk shows that one is the annihilation operator for particles and the other for antiparticles.

Which is higher energy red or violet?
The energy of a blue photon is equal to (6.62 x 10-34) x 1015 Hz i.e. 6.62 x 10-19 joules.

This is simply based on the optical properties of light. A photon is no smaller than what can be achieved with an optical beam, because a photon is just the lowest energy state of the field (above the vacuum level). The laws of nature are very nearly symmetrical with respect to particles and antiparticles. For example, an antiproton and a positron can form an antihydrogen atom, which is believed to have the same properties as a hydrogen atom. This leads to the question of why the formation of matter after the Big Bang resulted in a universe consisting almost entirely of matter, rather than being a half-and-half mixture of matter and antimatter.
The photon model accounts for anomalous observations, including the properties of black-body radiation, that others (notably Max Planck) had tried to explain using semiclassical models. In that model, light is described by Maxwell's equations, but material objects emit and absorb light in quantized amounts (i.e., they change energy only by certain particular discrete amounts).
An electron can be bound to the nucleus of an atom by the attractive Coulomb force. A system of one or more electrons bound to a nucleus is called an atom.
What is a photon easy definition?
A photon is the quantum of electromagnetic radiation that describes the particle properties of an electromagnetic wave. The energy of a photon is given by the equation E = hf, where E is energy, h is Planck's constant, and f is frequency.