$ 0.253 1.15%
RChain (RHOC) Rank 3864
| Mkt.Cap | $ 0.00000000 | Volume 24H | 0.00000000RHOC |
| Market share | 0% | Total Supply | 870.66 MRHOC |
| Proof type | N/A | Open | $ 0.25 |
| Low | $ 0.25 | High | $ 0.26 |
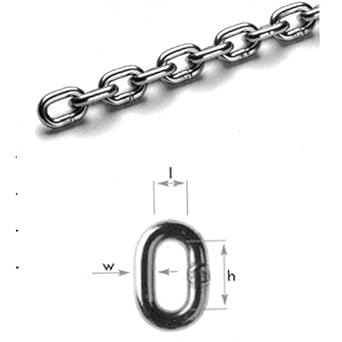
Finding the Right Saw Chain for You
@rchain_coop
Strictly speaking, it doesn’t—it turns out that the isolated β-subunit can create ATP from ADP and ionic phosphate all by itself. In doing so, the β subunit adopts two distinct conformational states. Once both are bound, β transitions to the second state, which binds ATP with exceptionally high affinity but no longer binds ADP and phosphate. Formation of several avid but non-covalent bonds between β and ATP provides the energy that is needed to create the new, energy-rich phosphate anhydride bond in ATP. The interaction of the rotating γ subunit with the static β subunits is crucial to ATP synthesis.

Due to the complexity of the project and disagreement between team members, it was decided that Greg would focus on realizing the vision of RChain. The RChain organizations (the Co-op and Holdings company) were formed in December January 2017.
Therefore, to make electrons flow along the prescribed path along complexes I–IV, these proteins must provide functional groups that are able to accept and donate electrons. These groups must be closely spaced, within a few Angstroms of each other, to allow for efficient electron transfer. Furthermore, to persuade the electrons to flow in the right direction, the successive transitions must be exergonic, that is, their free energy (ΔG) must be negative. The workings of the respiratory chain are quite different from all other pathways in human metabolism. Each of those other pathways consists of a succession of discrete enzymatic reactions.
In the next frame, the disk has moved a bit and accepted the proton; it then goes around by almost a full turn, until it reaches the other conduit (bottom right). In the final frame, the proton has left the disk and is on its way to the mitochondrial matrix, while the c subunit is about to move on and pick up then next proton. chain has a strategic aspartate residue that faces the surrounding lipid membrane and can reversibly accept a proton.

The amount of ATP given in the preceding slide is a theoretical maximum. In reality, the ATP yield will be significantly lower, because some protons are used for purposes other than driving ATP synthase. ATP synthesized in the mitochondrion must be exported to the cytosol, and ADP produced there has to get back in. This is accomplished by a special transporter protein in the inner mitochondrial membrane that exchanges ATP and ADP for each other. Since ATP carries one more negative charge than ADP (ATP4– vs. ADP3–), this exchange amounts to a net export of one negative charge, or to the net import of one positive charge per ATP.
Protons and other ions can flow across a salt bridge between the chambers so as to preserve electroneutrality. In order to allow the flow of ions but prevent mixing of the chamber contents by convection, this hole is covered with a porous membrane or plugged with agar. The synthetic compound dinitrophenol can diffuse across the inner mitochondrial membrane in both its protonated and unprotonated form. It can therefore carry protons into the mitochondrion, thereby dissipating the driving force for ATP synthase.
The two mitochondrial isocitrate dehydrogenases
Powered by this electron migration, complex I ejects four protons across the membrane. allow the observation of electron transport without ATP synthesis. On the other hand, ATP synthesis works without electron transport if a proton gradient is created in some other way. The pyrrole rings or sulfur atoms do not only keep the iron ions in place but also modulate their redox potentials. These potentials are further tweaked by the specific molecular environment of each cofactor, in such a way as to enable the electrons to flow from one cofactor to the next.

Business English
Because of this clear distinction, we can safely examine these two functions separately from each other. The respiratory chain involves four large protein complexes (I–IV) as well as ATP synthase (AS). Coenzyme Q (Q) and cytochrome C (C) are diffusible electron carriers.
RChain
The basic idea is that capture and release of electrons cause conformational changes to a protein. This is entirely analogous to conformational changes caused by allosteric effectors binding to enzymes, or by phosphate groups bound to cytoskeletal proteins such as the myosin light chain. Since cytochrome C delivers the electrons to complex IV one at a time, the reduction will involve partially reduced oxygen species. As noted above, partially reduced oxygen species are reactive and toxic when let loose upon the cell, so their premature release from cytochrome C oxidase must be minimized. , complex III contains several more redox co-factors that act as intermediate stepping stones in the electron transfer steps outlined above.

- Due to the complexity of the project and disagreement between team members, it was decided that Greg would focus on realizing the vision of RChain.
- are based on the assumption that complexes I, III, and IV pump 4, 2, and 4 protons, respectively.
- Where protons are stripped off, they may be preferentially released at the cytosolic side, whereas uptake of protons may preferentially occur on the mitochondrial side.
- chain has a strategic aspartate residue that faces the surrounding lipid membrane and can reversibly accept a proton.
It is commonly stated that approximately ten protons are ejected for each pair of electrons abstracted from NADH, such that four protons are ejected at each of complexes I and IV, and 2 at complex III. As electrons are withdrawn from the solute in one chamber and delivered to the other, the voltmeter indicates the direction and magnitude of the potential difference.
FSC homepage

Otherwise, the different subunit stoichiometry of F0 should directly translate into a different ATP yield in the entire respiratory chain. Therefore, complex III uses ubiquinone as a prosthetic group to facilitate the movement of protons across the membrane.
Iron and copper are subsequently restored to their original oxidation levels further electrons obtained from cytochrome C. The iron sulfur cluster donates its electron to cytochrome C, while the heme transfers its electron to the second molecule of ubiquinone in site B. The ubiquinone molecule in site A, now oxidized, trades places with another one in the surrounding membrane that was reduced in the preceding steps of the respiratory chain. As an example of of the foregoing, we will have a look at the (in)famous ubiquinone cycle. Ubiquinone (or coenzyme Q) is a hydrogen carrier; like FAD and FMN, it can carry two electrons but can accept and donate them one at a time.
Chain Anatomy - Parts of a Cutter
Moreover, ionic phosphate produced by cytosolic ATP cleavage also must return to the mitochondrion, which consumes another proton. These processes detract considerably from the theoretical efficiency of ATP synthesis.
and coenzyme Q carry both electrons and protons—that is, hydrogen. In contrast, the hemes and the iron-sulfur clusters carry only electrons. The flavin nucleotides flavin adenine dinucleotide (FAD) and flavin mononucleotide (FMN) can occur in three different states of reduction, which differ by single electrons. Unlike NAD, which can accept or donate electrons only in pairs, flavins can accept or yield electrons one at a time. Therefore, flavins can buffer the electron flow between NAD and iron-containing redox cofactors, and this is why the very first cofactor that accepts the electrons from NADH in complex I is indeed an FMN molecule.
In particular, it cuts out oxaloacetate, whose low concentration inside the mitochondrion probably forms the kinetic bottleneck of the malate-aspartate shuttle. It is interesting to note that the glycerophosphate shuttle is highly active in insect muscle, which has an extremely high ATP turnover during flight. , in keeping with its position at the start of the transport chain. It has a slightly higher potential than NADH and is therefore able to accept its electrons.

This means that electrons are stripped of their protons and rejoined by protons again repeatedly during transport. Where protons are stripped off, they may be preferentially released at the cytosolic side, whereas uptake of protons may preferentially occur on the mitochondrial side. This would account for some, but not all of the proton translocation occurring in the respiratory chain. In the glycerophosphate shuttle, the hydrogen is never actually transported to the mitochondrion. Dihydroxyacetone phosphate serves as the intermediate hydrogen acceptor and is reduced in the cytosol to glycerophosphate by glycerophosphate dehydrogenase.

This cofactor receives an electron from another cofactor upstream (A), which then becomes occupied by the next electron (B). Electrostatic repulsion moves the valve (C), such that the proton is translocated and the electron can jump to the next cofactor downstream (D). The extended spring—which is a metaphor for conformational strain—then returns the valve to its original position (E).
, you will notice that the difference in the redox potentials of FAD and ubiquinone is rather small. Consequently, the amount of free energy associated with the transfer of electrons from FAD to ubiquinone is too small to permit the performance of work against the proton gradient. and coenzyme Q, which means that very little energy is released as electrons traverse this complex. Such minor steps in redox potential suffice to jog the electrons along, but they are too small to contribute to proton pumping. This step in potential corresponds to a significant amount of free energy that is released at some point within complex I as the electrons travel through it from FMN toward N2.

This slide shows the structures of the four respiratory chain complexes that form the respiratory chain. In this scheme, the mitochondrial matrix is below the membrane, whereas the cytosol is above it.
subunit are embedded in the inner mitochondrial membrane, whereas the other subunits of the molecule protrude into the mitochondrial matrix. indicates a contribution of only 6 kJ/mol from the concentration gradient. In summary, therefore, the proton-motive force is dominated by the membrane potential. In our conceptual model, a spring-loaded valve controls the proton conduit. The lever of the valve carries an electron-transporting cofactor (a heme or FeS-cluster).







